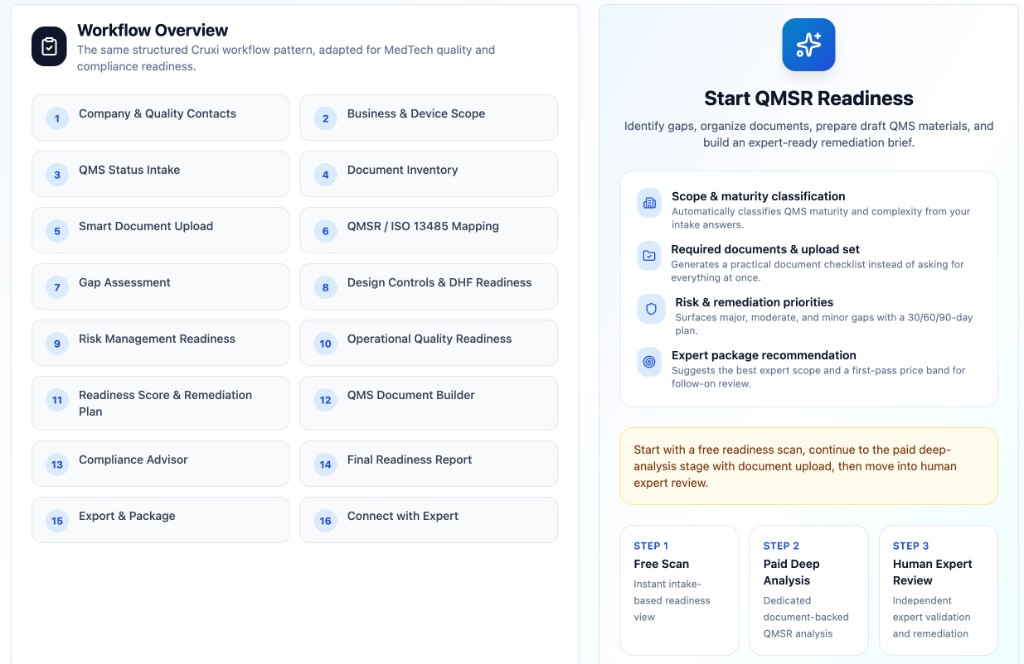
Start free. Go deeper only when needed.Cruxi helps medical device teams assess QMSR / ISO 13485 readiness in three steps: (1) free scan for early visibility, (2) paid deep analysis for document-based findings, (3) optional expert review for validation and remediation. Because Cruxi handles the heavy first pass, consultants can spend less time on discovery and more time on what matters.
QMSR / ISO 13485 Readiness Assessment for Medical Device Teams
Free QMSR readiness scan · Paid deep analysis · Optional expert review
Start with a free scan to identify likely QMS gaps. Upgrade to deep document analysis for evidence-based findings, then add expert review only if you need validation or remediation support.
Free scan
Assess QMS maturity, likely gaps, and missing documents from structured intake.
Paid deep analysis
Upload documents for detailed gap analysis, readiness scoring, and a structured roadmap.
Optional expert review
Bring in a consultant after Cruxi has organized materials and surfaced likely issues.
Preliminary readiness assessment only. Not an FDA inspection, ISO certification audit, legal opinion, or substitute for qualified expert judgment.
Free scan first; paid deep analysis when you want document-backed outputs. Typical guided effort for the full path: about 2–4 hours.
How it works
Three clear stages: free entry, paid document work, optional human validation—so you only pay for depth when it makes sense.
Free scan
FREE: Start with a free readiness scan to understand your QMS maturity, likely gaps, and missing documents from intake—before you commit to deeper work.
Paid deep analysis
PAID: Upgrade to deep analysis to review uploaded QMS documents, identify evidence-backed weaknesses, and generate a structured readiness package you can use internally or hand to a consultant.
Optional expert review
OPTIONAL EXPERT: Add expert review only when needed, after Cruxi has already organized materials and surfaced likely issues—for validation, prioritization, or remediation support.
Ready for document-backed outputs? Upgrade to Deep Analysis in the app when you are.
Why this can reduce expert fees
VALUE: Because Cruxi does the heavy first pass, consultants can spend less time on discovery and more time on judgment, remediation, and implementation. That can reduce wasted consultant hours and make expert support more efficient. Cruxi does not guarantee savings; scope and rates depend on your provider and project.
- Cruxi organizes your QMS materials before consultant review, which can reduce discovery time.
- Structured analysis surfaces likely missing SOPs, weak procedures, and document gaps first—helping avoid paying experts mainly for basic sorting and initial gap mapping.
- Consultants can start from a structured readiness package instead of messy folders—may lower consultant cost for early-phase work.
- Less paid discovery time means expert hours can go to validation, remediation sequencing, and implementation.
- Better organization upfront may reduce unnecessary consulting cost—especially when you only engage experts after the package is prepared.
Free scan vs paid deep analysis vs expert review
What you get at each stage. Exact depth depends on your intake and uploads; this table summarizes the offer ladder.
| Free scan | Paid deep analysis | Expert review (optional, later) | |
|---|---|---|---|
| Intake review | Yes (core of free stage) | Yes (extends with your answers) | Yes (validates your package) |
| Document upload & evidence review | Limited / optional (early visibility) | Yes (document-backed work) | Uses your prepared package |
| Readiness score | Orientation-level | Document-informed | — |
| Missing document checklist | Yes (likely gaps) | Yes (refined with uploads) | — |
| Evidence-based findings | Limited (intake-led) | Yes | — |
| Gap matrix | Simplified / directional | Yes | — |
| Remediation roadmap | High-level | Yes (e.g. 30/60/90-day) | May finalize priorities |
| Draft SOPs / templates | — | Yes (drafts for qualified review) | May refine |
| Consultant validation | — | — | Yes |
| Implementation support | — | — | Optional (independent consultant) |
Cruxi does not certify your QMS, provide legal advice, or determine FDA inspection outcomes. Expert review is recommended before relying on outputs for certification, inspection, launch, or diligence.
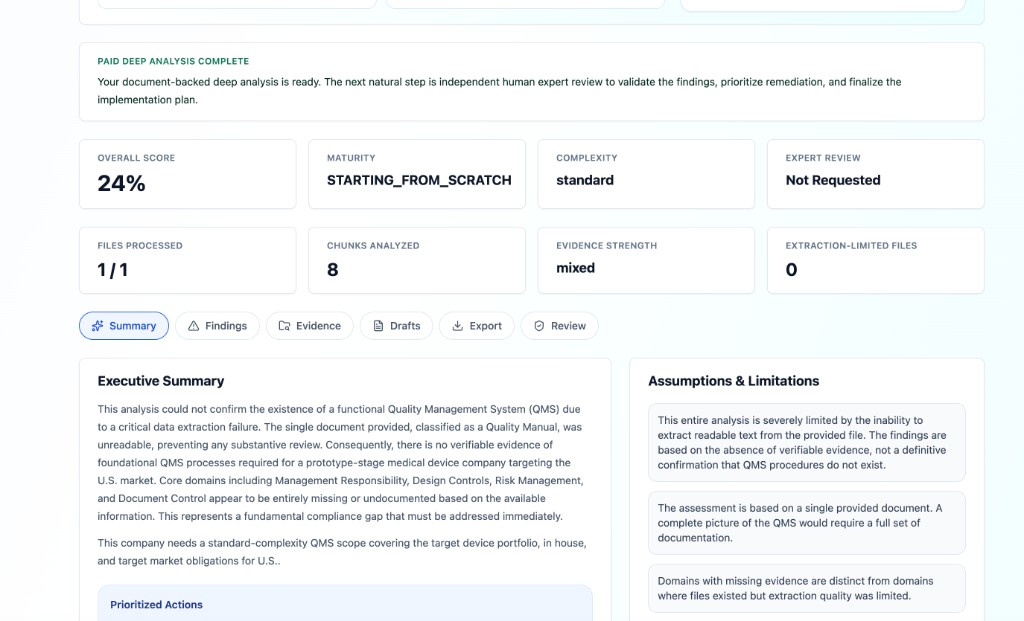
What you get in paid deep analysis
After the free scan, upgrading unlocks document-backed deliverables: scores, matrices, checklists, maps, roadmap, and drafts your team—or a consultant—can act on. See the comparison table above for free vs paid vs expert.
Readiness score
Prioritized view of QMS maturity and gap severity for your scope.
Gap matrix
Area-by-area coverage: strong, partial, missing, or not applicable.
Missing-document checklist
SOPs, records, and evidence still needed for your stage.
Design controls / DHF gap map
Traceability and DHF completeness vs. common expectations.
Risk management gap map
Risk file, controls, verification links, and post-market hooks.
30/60/90-day roadmap
Practical remediation sequence you can execute or hand to an expert.
Draft SOP bundle
First-draft procedures and templates for qualified review.
Expert-ready brief
Structured package so review starts from evidence, not blank discovery.
Later-stage only: Add Expert Review after your AI package is prepared.
Who this is for
Teams that need a clear QMSR / ISO 13485 gap picture before the next regulatory or business milestone.
Startups building a QMS
Turn scattered drafts into a prioritized build sequence and document list.
Teams preparing for FDA submission
Stress-test design history, risk files, and core procedures against common review expectations.
Companies preparing for manufacturing or launch
Close gaps in production, supplier, training, CAPA, and complaint handling before scale-up.
ISO 13485 companies checking QMSR alignment
Map certificate-level documentation to FDA QMSR framing and evidence needs.
Foreign manufacturers entering the U.S.
Clarify U.S. market QMS expectations and documentation gaps early.
Companies preparing for diligence or remediation
Package findings for boards, buyers, or post-audit remediation with less discovery churn.
- QMSR effective February 2, 2026. FDA's rule incorporates ISO 13485:2016 by reference.
- Assessment logic is aligned to ISO 13485:2016-based readiness patterns—not a substitute for certification or inspection.
- Findings are tied to your intake responses and uploaded documents, with traceable gap notes.
- Optional expert review—add later for validation; not a substitute for certification or inspection.
Upgrade to Deep Analysis when you are ready for document-backed outputs.
QMSR context
The same guided path runs from free scan through paid deep analysis; expert review is optional and later. Your quality owner stays in control at every stage.
FDA's revised Part 820 is the Quality Management System Regulation (QMSR), effective February 2, 2026, incorporating ISO 13485:2016 by reference for medical device QMS expectations.
Materials stay in Cruxi unless you authorize sharing for a quote or review. Outputs are not certification, legal advice, or an FDA inspection determination.
Add Expert Review — optional, after your package is ready
Regulatory disclaimer
This is a preliminary QMSR / ISO 13485 readiness assessment based on your intake and documents. It is not an ISO certification audit, FDA inspection determination, legal opinion, or guarantee of compliance. Expert review is recommended before relying on outputs for formal regulatory or commercial milestones.
What Cruxi assesses
Intake covers these areas in the free scan; paid deep analysis adds document-backed depth across the same scope.
Company & Quality Scope
Primary quality owner, regulatory contact, management representative, manufacturing role, target markets, and provider coordination details.
Device type, device stage, manufacturing model, outsourced partners, markets, device complexity, and QMS scope.
Current QMS maturity, ISO 13485 certification status, eQMS usage, audit history, inspection history, open CAPAs, complaints, and existing procedures.
Document & Process Readiness
Identify which SOPs, records, forms, logs, DHF artifacts, risk files, supplier records, audit reports, and management review materials exist.
Cruxi requests the most important documents first instead of asking for your entire QMS folder.
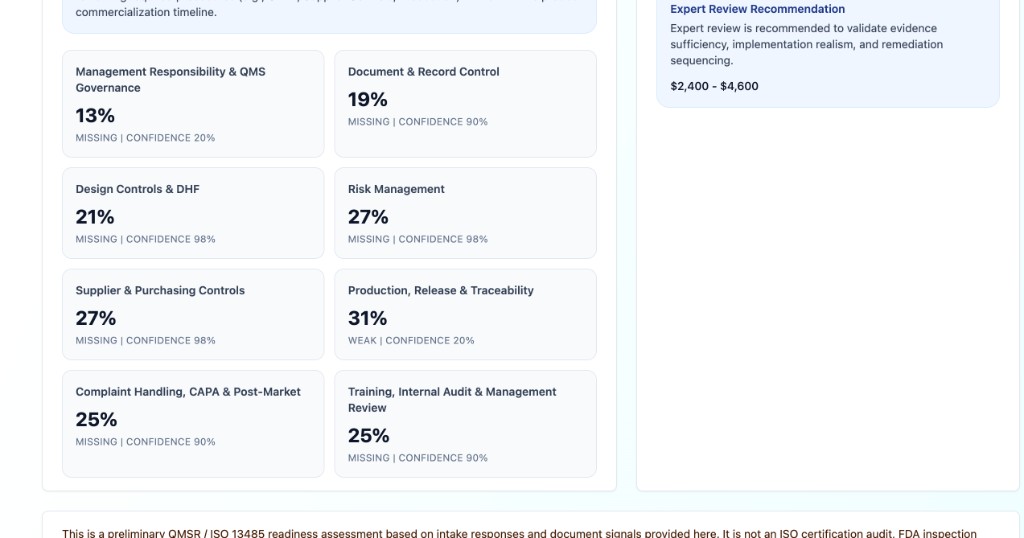
Cruxi maps your documents and intake responses against core QMS areas including document control, management review, design controls, risk management, CAPA, complaints, supplier controls, training, production controls, and records.
Gap Analysis & Remediation
Find missing procedures, generic templates, weak SOPs, missing records, undefined responsibilities, and lack of implementation evidence.
Review user needs, design inputs, outputs, verification, validation, design reviews, traceability, DHF index, and design-transfer gaps.
Assess risk-management plan, hazard analysis, risk controls, verification links, residual risk, benefit-risk rationale, and post-market risk monitoring.
Review CAPA, complaints, supplier qualification, training, internal audit, management review, nonconformance, change control, labeling control, and production-release processes.
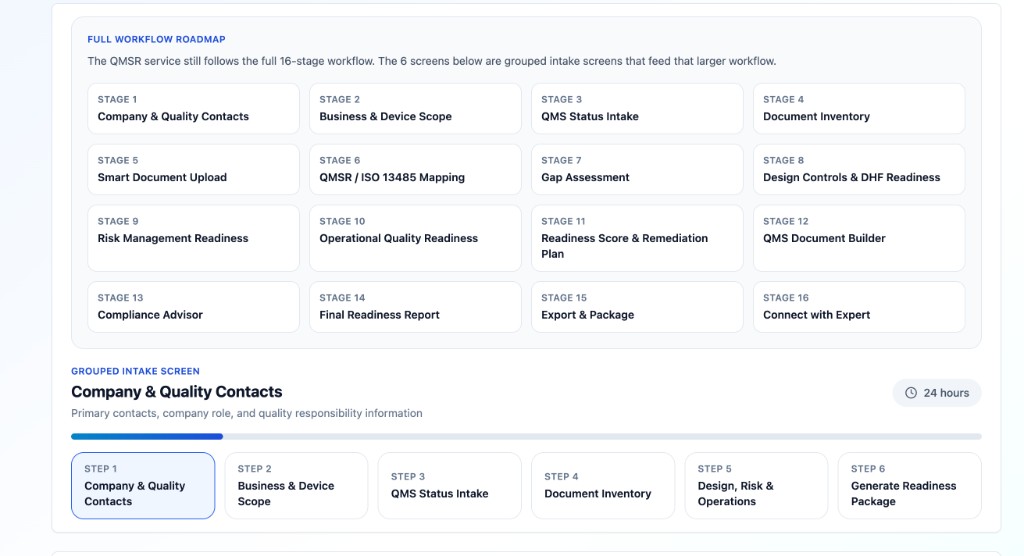
One workflow: free scan → deep analysis → optional expert
After you upgrade to paid deep analysis, one guided path covers scope, documents, core QMS processes, DHF, risk file, and operational quality—then packages scores, matrices, drafts, and an expert-ready brief. You do not need separate “modules” to get a full QMSR / ISO 13485 gap picture.
- assignment_turned_inQMS maturity, missing SOPs, records, and implementation evidence
- device_hubDesign controls / DHF traceability and completeness
- warningRisk file structure, controls, verification links, residual risk, post-market monitoring
- buildCAPA, complaints, suppliers, training, audits, production and release readiness
- descriptionDraft SOPs, forms, and logs sized to your company and device profile
Assistive analysis, your oversight
Cruxi automates structured intake, document organization, and gap detection. It does not certify your QMS, act as an FDA inspector or ISO auditor, or provide legal advice.
- rate_reviewFindings are preliminary—have your quality owner or consultant validate before formal use.
- linkGaps reference your intake answers and uploads where possible.
- lockYour project materials are not used to train public models.
Frequently Asked Questions
Free scan, paid deep analysis, optional expert—and how that can make consultant time more efficient.
What is included in the free scan?
The free scan is intake-led: you get early visibility into QMS maturity, likely gaps, and missing documents from structured questions. It is not a full document-backed review—that is what paid deep analysis is for.
What do I get with paid deep analysis?
You upload QMS documents for evidence-backed review. Outputs typically include a more informed readiness score, gap matrix, missing-document checklist, evidence-based findings, remediation roadmap, draft SOPs or templates (drafts for qualified review), and a structured package you can share internally or with a consultant—depth depends on what you upload.
When should I add expert review?
Add expert review when you want independent validation, help prioritizing remediation, or implementation support—usually after Cruxi has organized materials and produced a structured package. It is optional and does not compete with starting free.
Can this reduce consulting cost?
Cruxi cannot guarantee lower expert fees. By organizing documents and running a structured first-pass gap analysis before you engage consultants, it can reduce discovery work and help avoid paying experts primarily for basic document sorting and initial gap mapping—making engagement more efficient. Actual savings depend on your provider, scope, and project.
Can I start free and decide later?
Yes. Start with the free scan, then upgrade to paid deep analysis only if you want document-backed findings and a structured readiness package. Add expert review later if needed.
What is QMSR?
QMSR is FDA's Quality Management System Regulation for medical devices. It became effective February 2, 2026, and incorporates ISO 13485:2016 by reference into FDA's device QMS framework.
Is ISO 13485 certification required by FDA?
No. QMSR aligns with ISO 13485:2016 concepts, but FDA does not treat an ISO certificate as a substitute for FDA requirements or inspection.
Does Cruxi certify my QMS?
No. Cruxi is not a certification body, notified body, FDA inspector, or law firm. Outputs are assistive; qualified experts should validate before formal reliance.
What documents should I upload for paid deep analysis?
Prioritize: Quality Manual, SOP index, Document Control, Design Control, Risk Management (SOP or risk file), CAPA, Complaints, Supplier Control, Training, plus any recent audit or gap report. Partial uploads still help; more complete files sharpen evidence-based findings.
Is this useful if we are starting from scratch?
Yes. The free scan still orients you; paid deep analysis can produce a prioritized build list, drafts for review, roadmap, and expert-ready brief even when documentation is thin.
Start free. Upgrade when you need document depth.
Use the free scan for orientation, paid deep analysis for evidence-backed gaps and a structured package, and add expert review only if you want validation or remediation help—after the heavy discovery is already done.
Not certification, legal advice, or an FDA inspection determination. Expert judgment is recommended before formal reliance.